meisel2012failure
Failure of Adaptive Self-Organized Criticality during Epileptic Seizure Attacks
Christian Meisel, Alexander Storch, Susanne Hallmeyer-Elgner, Ed Bullmore and Thilo Gross
PLoS Comput Biol 8, e1002312, 2012
Critical dynamics are assumed to be an attractive mode for normal brain functioning as information processing and computational capabilities are found to be optimized there. Recent experimental observations of neuronal activity patterns following power-law distributions, a hallmark of systems at a critical state, have led to the hypothesis that human brain dynamics could be poised at a phase transition between ordered and disordered activity. A so far unresolved question concerns the medical significance of critical brain activity and how it relates to pathological conditions. Using data from invasive electroencephalogram recordings from humans we show that during epileptic seizure attacks neuronal activity patterns deviate from the normally observed power-law distribution characterizing critical dynamics. The comparison of these observations to results from a computational model exhibiting self-organized criticality (SOC) based on adaptive networks allows further insights into the underlying dynamics. Together these results suggest that brain dynamics deviates from criticality during seizures caused by the failure of adaptive SOC.
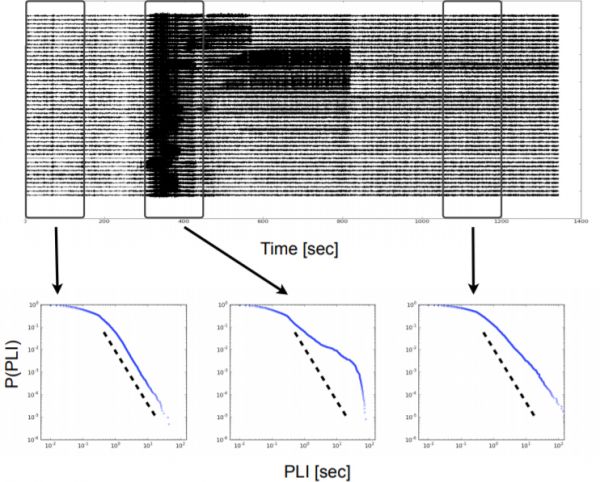
Figure 1: The distribution of phase-locking intervals deviates from a power-law during epileptic seizure attacks. Top: The
electrocorticogram (ECoG) recording shows the onset of a focal epileptic seizure attack around 300 seconds time. Bottom: Cumulative distributions of phase-locking intervals (PLI) are obtained during three time intervals of 150 seconds: pre-ictal (left), ictal (middle) and post-ictal (right). Dashed lines indicate a power-law in the double logarithmic plot. While the distribution appears to follow a power-law during the preictal period, intervals of increased phase-locking disturb this characteristic distribution with the onset of seizure activity. Data shown are from patient 1 at scale 3, corresponding to the frequency band 25-12.5 Hz.